 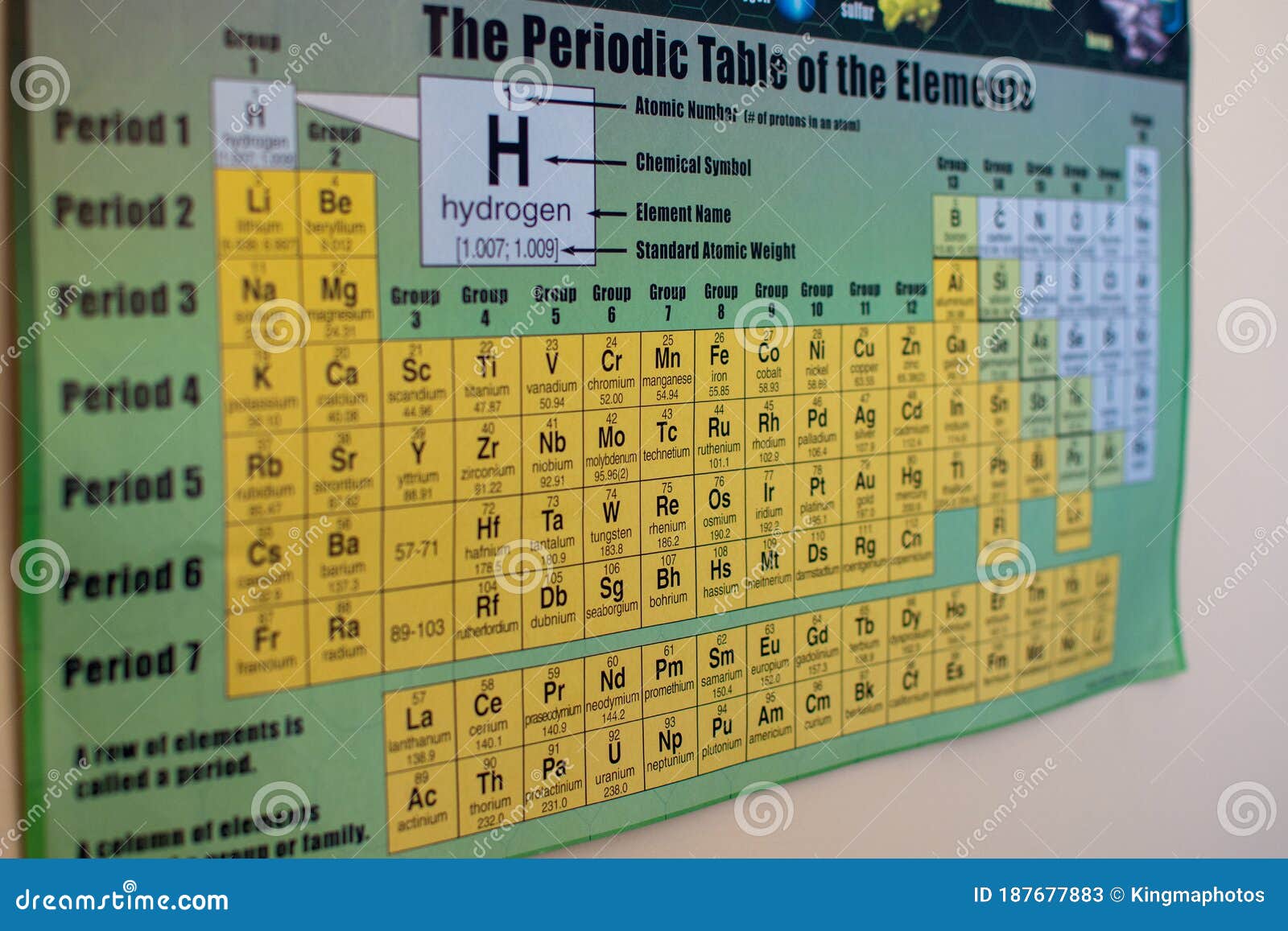
Sodium - Although abundant on Earth, sodium is never naturally found in its pure form it forms compounds with other elements.Įlements are the building blocks of life and human innovation.Oxygen - Oxygen is the third most abundant element in the universe however, its reactivity made it relatively rare in Earth's early atmosphere.Neon is the 5th most common element in the universe but comprises only 0.0018% of the Earth's atmosphere. It doesn’t respond with oxygen, helium, neon, and argon. Fluorine - Fluorine is the most receptive and electronegative of all the chemical elements.The iron and steel industries depend on it. charcoal from wood and coke from coal) is used in metal smelting. Carbon - The amount of carbon on Earth is relatively constant it’s simply transformed from one form to another by the carbon cycle.Lithium chloride is one of the most hygroscopic (absorbs moisture from the air) materials known and is used in air conditioning and industrial drying systems (as is lithium bromide). Lithium - Lithium is the lightest of all the metals and can easily float on water.Hydrogen can be combined with carbon dioxide to make methanol or dimethyl ether (DME) which are important transport fuels. Hydrogen -Although it’s rare on Earth, Hydrogen is the most common element in the universe.Helium - Helium atoms are so light that they can escape Earth's gravity! Helium is used in rocket propulsion (to pressurize fuel tanks, especially those for liquid hydrogen, because only helium is still a gas at liquid-hydrogen temperature).Beryllium ceramics are used to focus and control eye surgery lasers. Beryllium - Beryllium is two-thirds the density of aluminum and has six times the specific stiffness of steel by weight.Here are just a few that your students will enjoy learning: There are so many fun facts to discover about the different elements. But those just scratch the surface of the 118 elements. Of course, they may be familiar with some elements like glittering gold, iron, oxygen, and the voice-altering helium. Your lesson plans help students discover how the things they see every day (including the face in the mirror) contain these common and unusual elements. The researchers say that the familiar items reduce stress and help students successfully build their base of chemistry knowledge.Do the budding chemists in your classroom have questions about the stuff that everything is made of? Their curiosity is understandable since everything in nature, from the clouds to the trees and the air we breathe, is made of elements. An increase in correct answers relating to the subject on midterm exams suggested that these periodic tables improved students' comprehension. Over 75% of students taught with this strategy identified it as somewhat, very or extremely useful. #Periodic table chemistry in focus how to#For example, a superhero table featured characters, such as Superman, Wonder Woman and the Green Lantern, prompting in-class discussions on how to group the icons by ability, strength, gender and other properties - just as the elements are arranged in the real periodic table based on their similarities. The pseudo periodic versions replaced elements with single items to demonstrate one or more concepts needed to understand the trends and layout of the real chemical one. #Periodic table chemistry in focus series#The team presented first-year chemistry students with a series of made-up, or pseudo, periodic tables with objects that they had likely encountered before, including fruits and nuts, superheroes, iPad apps and meats. #Periodic table chemistry in focus full#And rather than teaching the full details right away, the researchers wanted to focus on some of its key characteristics first, using a contemporary, engaging and multi-level strategy. Gregory Watson and colleagues reasoned that introducing the periodic table with familiar items could also help. Previous educational studies presented the table in different formats, such as crossword puzzles and cartograms, to address multiple types of learning styles. However, many university students consider it a daunting and difficult subject to master, prompting professors to seek new ways to engage their students and make its concepts more accessible. One of the fundamental topics taught in first-year undergraduate chemistry courses is the organization and layout of the periodic table of elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
